Investors
Notice of publication of the Annual Report 2018 and other documents (in Italian) for the Shareholders’ meeting on April 30th, 2019
Notice of publication of the Annual Report 2018 and other documents (in Italian) for the Shareholders’ meeting on April 30th, 2019 ▾
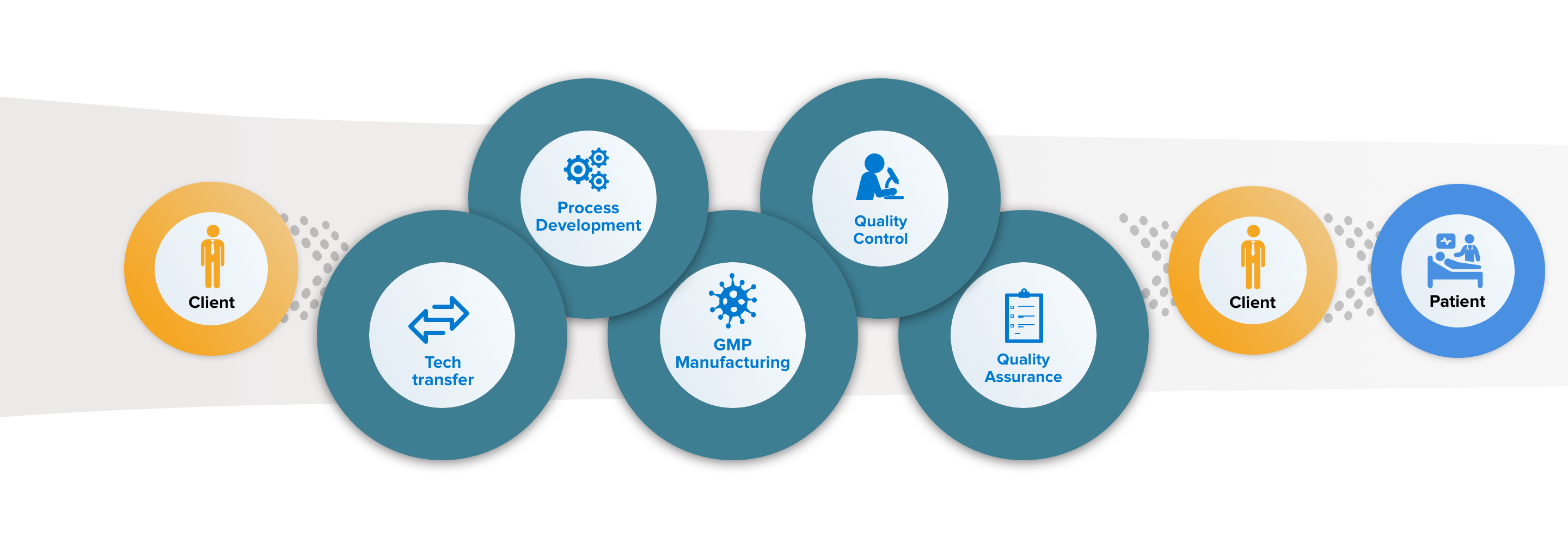
MolMed offers tailor-made services for cell and gene therapy projects, satisfying the customer's needs in terms of guaranteeing the highest clinical and commercial standards for ATMP.
Our services include technology transfer, development, scale up, automation and validation of RVV / LVV cell transduction processes, as well as the finalization and realization of the Medicinal Product. At the same time, MolMed offers the development, qualification and validation of analytical tests, both standard and product-specific.


Pipeline
Bresso Site_GMP
-
Surface area: about 3.300mq (35.000 SQF) divided in two floors
-
Designed and built in accordance with the highest quality standards
-
First Floor: 1.600mq (17.000SQF) dedicated to development, testing and quality control
-
Second Floor: 1.700mq (18.000SQF) dedicated to the production of viral vectors and Genetically modified cells according to cGMP
-
10 Grade B suites and 7 Grade C suites


