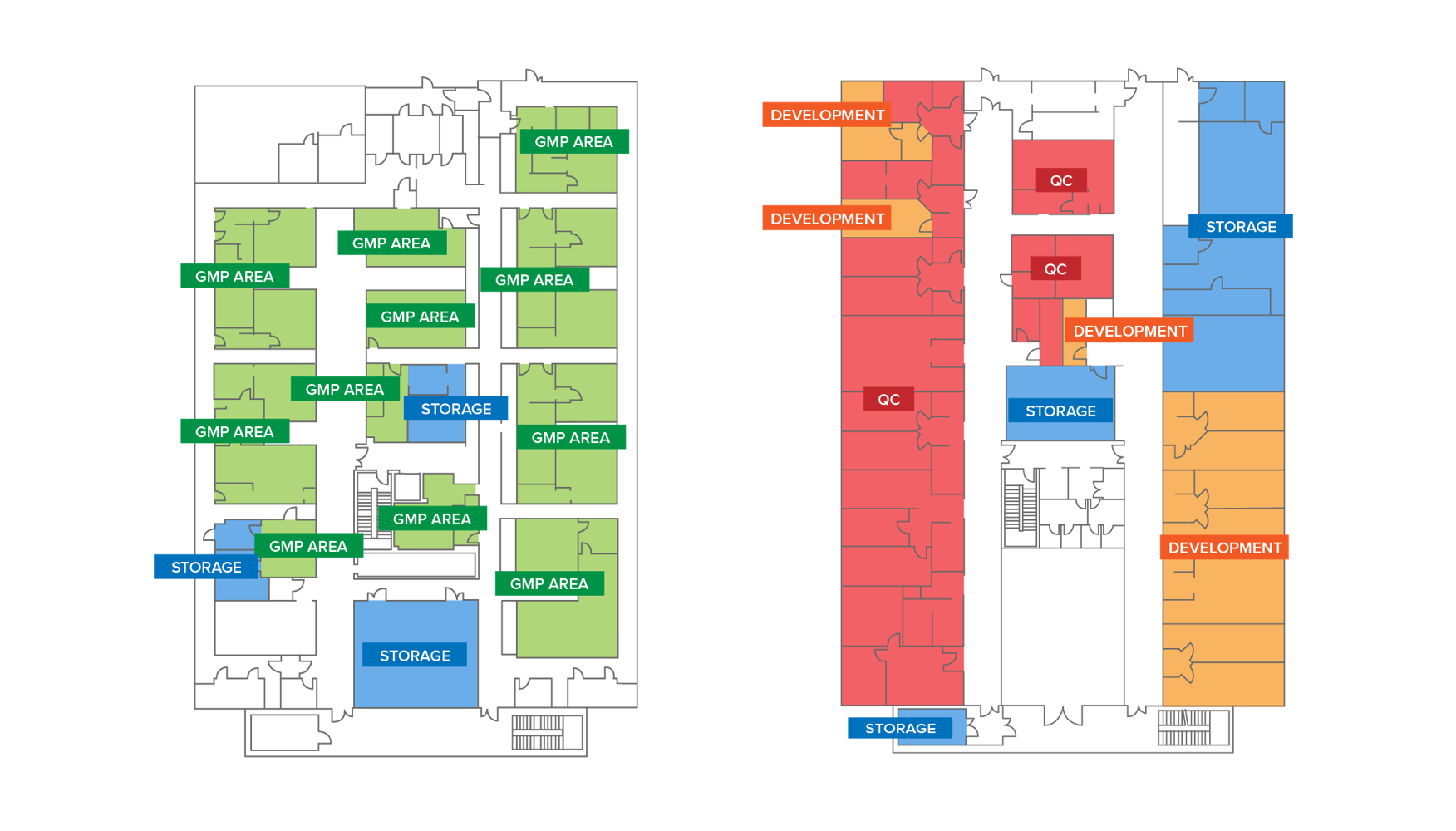
Bresso Site_GMP
-
Surface area: about 3.300mq (35.000 SQF) divided in two floors
-
Designed and built in accordance with the highest quality standards
-
First Floor: 1.600mq (17.000SQF) dedicated to development, testing and quality control
-
Second Floor: 1.700mq (18.000SQF) dedicated to the production of viral vectors and Genetically modified cells according to cGMP
-
10 Grade B suites and 7 Grade C suites
-
Authorization for the GMP production of viral vectors (on a 48L or 200L scale) and genetically modified cells for clinical trials and market